Primary non-Hodgkin lymphoma of the parotid gland: a case report
Article information
Abstract
Most malignant lymphomas of the head and neck region are non-Hodgkin lymphomas (NHL), and diffuse large B-cell lymphoma is the most common subtype. The prevalence of malignant lymphoma among parotid tumors is low, approximately 1% to 4%. The most common symptom of parotid lymphoma is a unilateral, non-tender, firm mass that slowly grows in size over time. As its clinical manifestations are non-specific, a comprehensive assessment is required for an accurate diagnosis. The initial work-up includes imaging tools, such as computed tomography and magnetic resonance imaging. However, NHL of the parotid gland is difficult to distinguish from other types of benign tumors prior to biopsy; histopathological evaluation and subsequent immunohistochemical staining are needed for the final diagnosis. Once a definitive diagnosis is established, patients should be referred to an oncologist for staging. Treatment is mainly based on systemic chemotherapy, whereas radiotherapy is indicated for certain cases. Here, we report the case of a 53-year-old man who presented with a progressively enlarging mass in the right parotid area, which was later diagnosed as malignant lymphoma of the parotid gland after superficial parotidectomy.
INTRODUCTION
Most parotid gland tumors are benign, and parotid gland cancers are relatively rare [1]. The most common benign neoplasm is a pleomorphic adenoma, followed by Warthin’s tumor. Mucoepidermoid carcinoma, cystic adenoid carcinoma, and squamous cell carcinoma are the three most frequent malignant neoplasms. Primary non-Hodgkin lymphoma (NHL) of the salivary gland is rare, accounting for approximately 2% of salivary gland neoplasms overall [2]. The most common site is the parotid gland (79%), followed by the submandibular gland (18%), minor salivary gland (2%), and sublingual gland (1%) [3]. As 80% of parotid gland tumors are benign, malignancies are usually not considered as differential diagnoses. Malignant lymphoma is often neglected in parotid masses. While the treatment of both benign and malignant tumors is mostly surgical, such as lumpectomy and parotidectomy, the treatment of lymphoma generally requires chemotherapy and/or radiotherapy. Thus, delayed diagnosis may lead to aggravation of the disease.
We report a case of extranodal, follicular subtype of parotid gland NHL. A 53-year-old man presented with a progressively growing parotid mass. It was initially suspected to be a benign tumor but was later diagnosed as malignant lymphoma after superficial parotidectomy.
CASE REPORT
A 53-year-old man presented with a slow-growing, non-tender, hard mass in the right parotid gland area (Fig. 1). The patient had no other symptoms or history of autoimmune disease or malignancy. Laboratory tests showed no abnormalities. The patient underwent contrast-enhanced computed tomography (CT), which displayed a 3.3×2.0-cm-sized homogenous mass with multiple enhanced nodules adjacent to the lesion (Fig. 2). CT suggested Warthin tumor, with a low possibility of a mixed tumor or low-grade mucoepidermoid carcinoma. Subsequent surgical resection was planned. Hence, superficial parotidectomy was performed through a preauricular incision, while preserving the left facial nerve and parotid duct. The excised specimen was approximately 5.7×3.0 cm in size and dumbbell-shaped (Fig. 3).
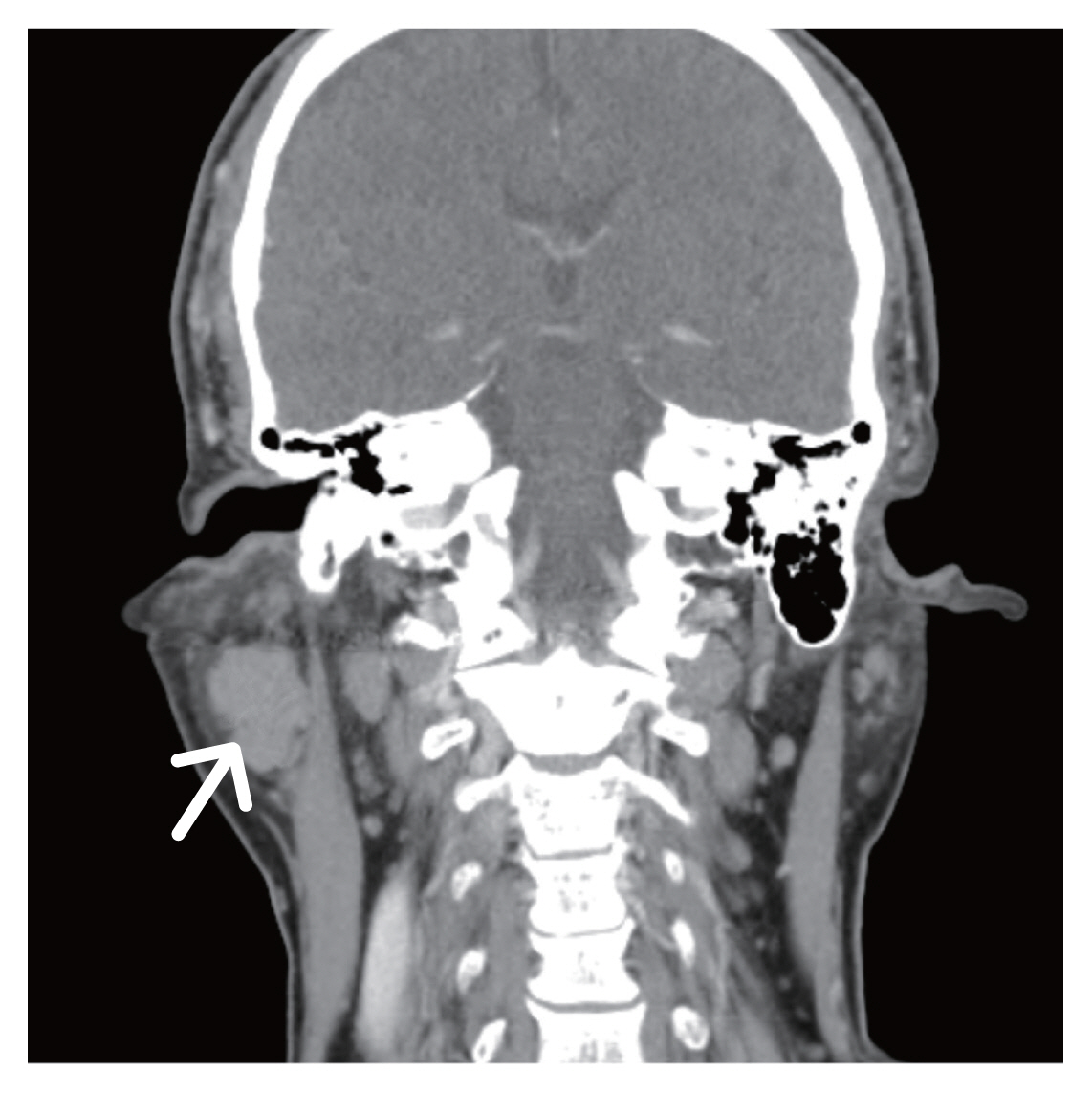
Preoperative computed tomography showing a 3.3×2.0-cm-sized enhancing homogeneous mass in the superficial lobe of the right parotid gland (white arrow).
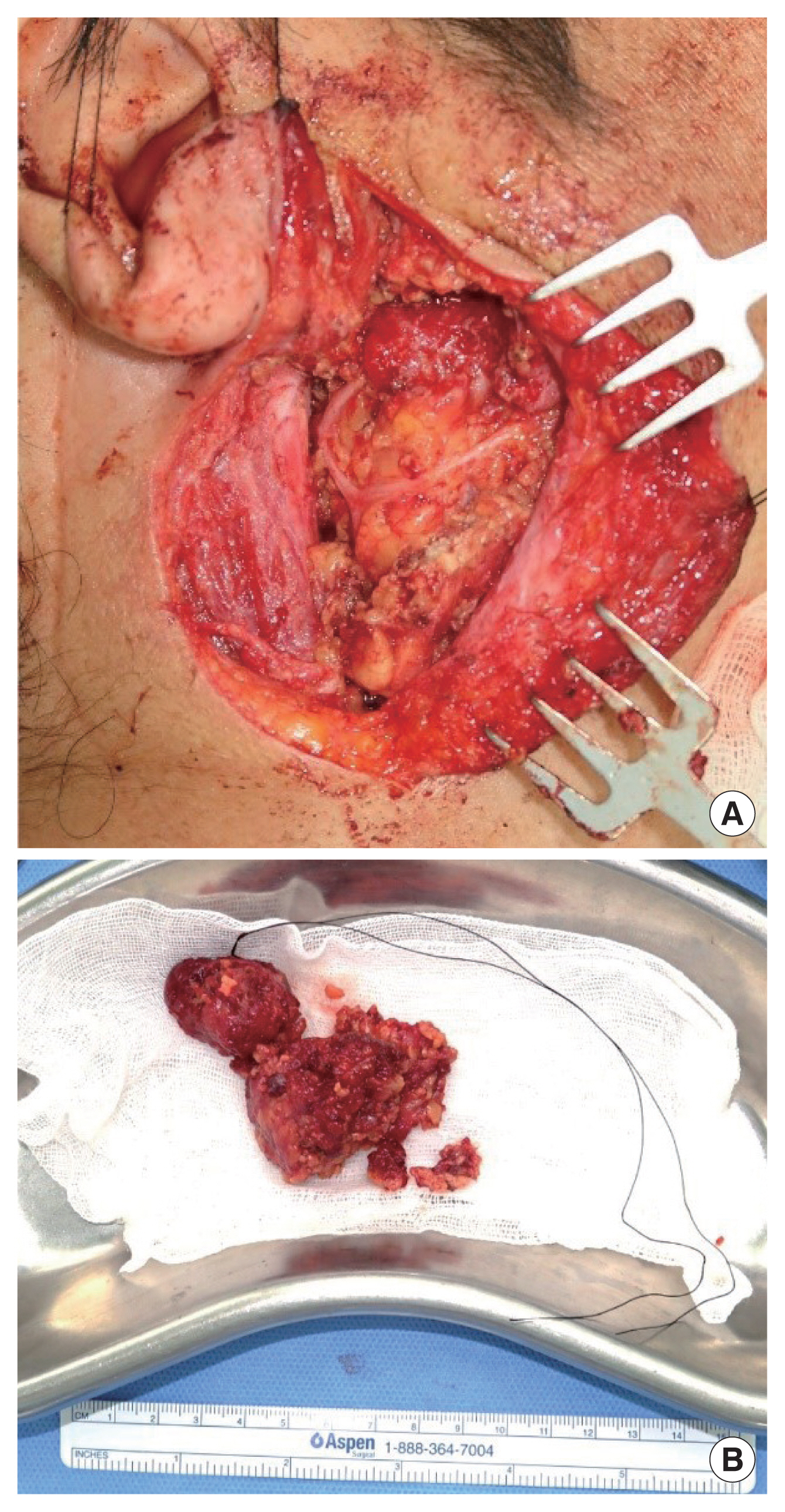
Intraoperative photographs. (A) After superficial parotidectomy with preservation of the facial nerve. (B) Dumbbell-shaped surgical specimen.
Microscopically, a population of small atypical lymphoid cells with hyperchromatic nuclei and scanty cytoplasm was observed using hematoxylin and eosin staining. On additional immunohistochemical (IHC) staining, malignant lymphoid cells were positive for CD20 and Bcl-2 proteins and negative for CD3, showing a low Ki-67 proliferative index (Fig. 4). Based on these IHC features, the overall results were consistent with grade 1–2 follicular lymphoma originating from B lymphocytes. The patient was referred to an oncologist for further evaluation and appropriate management. The positron emission tomography-CT revealed multiple pathological lymph nodes in cervical chains, axillae, and mesentery. Bone marrow examination showed bone marrow involvement. Accordingly, the final diagnosis was made as the Ann Arbor stage IVA. The patient underwent six cycles of chemotherapy with bendamustine and rituximab and did not receive radiation therapy. After chemotherapy, the patient achieved complete remission. The rituximab-based maintenance therapy was continued for 2 more years. The patient is currently under regular follow-up appointments with no signs of recurrence.
DISCUSSION
Although NHL is the second most common neoplasm in the head and neck region, primary parotid NHL accounts for only 1% to 4% of all parotid tumors [4]. The three most common subtypes of primary parotid NHL are extranodal marginal zone B-cell lymphoma of the mucosa-associated lymphoid tissue, follicular lymphoma, and diffuse large B-cell lymphoma, all of which originate from B lymphocytes [5]. Salivary gland lymphomas are frequently associated with a history of autoimmune diseases such as Sjögren syndrome [6]. The most common symptom is a progressively enlarging, non-tender solitary mass in the parotid area [7]. As the clinical signs of parotid lymphoma are non-specific, they cannot be easily distinguished from other benign or malignant tumors. Thus, malignant lymphoma is rarely suspected by physicians preoperatively.
The initial work-up usually begins with radiological examinations. CT is usually considered the modality of choice for the following reasons: first, it is cost effective; second, it is easily accessible; third, it provides a lot of information, such as tumor size, location, and adjacent structures; and finally, it yields rapid results [8]. Zhu et al. [9] described an ill-defined mass of relative homogeneity as the most common CT finding in follicular lymphomas. Other unusual features include a diffuse mixed solid cystic mass with an ill-defined margin and multiple solid masses with well-circumscribed borders. Magnetic resonance imaging (MRI) may also aid in the assessment of parotid neoplasms. According to previous literature, MRI findings of head and neck lymphoma generally display low signal intensity on T1-weighted images and low to high signal intensity on T2-weighted images with variable enhancement on gadolinium contrast [9]. King et al. [10] suggested that parotid lymphomas typically have a homogeneous intermediate T1 signal with an enhancing rim and a low T2 signal without the introduction of enhancement effects. However, the representative MRI findings of malignant lymphoma of the head and neck region remain controversial.
Definitive diagnosis of malignant lymphoma is confirmed based on histopathological evaluation combined with IHC staining [11]. Tissue samples can be obtained in several ways, including fine needle aspiration (FNA), core needle biopsy (CNB), and excisional biopsy, such as superficial parotidectomy. FNA plays a key role in evaluating salivary gland tumors but not necessarily in cases of malignant lymphoma [3]. Fakhry et al. [12] reported that the sensitivity and specificity of FNA for the diagnosis of malignancy are 80% and 89.5%, respectively. The high false-negative rate was particularly true for Warthin tumor, pleomorphic adenoma, and lymphoepithelial lesions. Since the diagnostic value of FNA can be influenced by insufficient cellularity, false-negative results may occur in malignant lymphoma [12]. Over the past few years, CNB has been increasingly preferred over FNA because of its higher sensitivity for diagnosing malignant tumors [13]. Novoa et al. [14] performed a prospective study of 103 patients with salivary gland neoplasms to compare the diagnostic accuracies of FNA and CNB. The sensitivities of CNB and FNA were 94% and 64%, respectively. The specificities of CNB and FNA were 100% and 95%, respectively. The results indicated that CNB was superior to FNA in detecting malignancies. However, there is still the risk of misdiagnosis. Small biopsy specimens obtained by CNB may not capture key lesions. As targeting errors can lead to an uncertain diagnosis, physicians should perform partial lobectomy to eradicate the risk of misdiagnosis due to inadequate tissue sampling.
Recent studies have also highlighted the usefulness of positron emission tomography-CT combined with 18F-labeled fluorodeoxyglucose (18F-FDG) as an initial staging tool. 18F-FDG is a biomarker of glucose metabolism and acts as an indicator of both structural and functional metabolism to identify the tumor burden. 18F-FDG avidity varies depending on the histologic subtype of the lymphoma. Based on its molecular characteristics and biological behavior, lymphoma is divided into two groups: aggressive and indolent. As noted by Wang et al. [15], aggressive lymphomas are highly avid on 18F-FDG, whereas indolent lymphomas show lower 18F-FDG avidity, consequently reducing the detection rate. Since the diagnostic accuracy of indolent NHL is not reliable, whole-body MRI has been suggested as an ancillary imaging test for the assessment of indolent lymphomas with low 18F-FDG avidity.
In this case, CT findings were highly suggestive of Warthin tumor, with a low likelihood of a mixed tumor and mucoepidermoid carcinoma. Hence, MRI or other invasive tests were not required for further evaluation. With the clinical impression of Warthin tumor, superficial parotidectomy was performed for both diagnosis and treatment. Once the final diagnosis and stage of lymphoma were established, the patient underwent chemotherapy. As shown in this case, since patients with parotid tumors are typically taken to surgery without a tissue diagnosis, the treatment of parotid lymphoma usually begins with initial surgical excision. Although the standard treatment of lymphoma is chemotherapy and radiation therapy, surgery does also play a role. Feinstein et al. [8] conducted a large cohort study of 2,140 patients, reporting that patients undergoing surgery were found to have a 35% reduced risk of mortality compared to those who did not undergo surgery.
When physicians encounter patients with parotid tumors, malignant lymphoma should be considered in the differential diagnosis. As NHL of the parotid gland is difficult to distinguish from other benign masses, we recommend utilizing different diagnostic tools when initial examination provides insufficient information. Although the validity of the surgical approach has been questioned, we argue that surgery encompasses clinical significance in the context of both diagnosis and treatment. First, surgical excisional biopsy allows prompt and precise diagnosis of lymphoma. Second, the treatment protocol—including surgery—can be an option since it may produce a satisfactory result when combined with radiotherapy and chemotherapy.
Abbreviations
CNB
core needle biopsy
CT
computed tomography
FNA
fine needle aspiration
IHC
immunohistochemical
NHL
non-Hodgkin lymphoma
MRI
magnetic resonance imaging
18F-FDG
18F-labeled fluorodeoxyglucose
Notes
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Ethical approval
This study received exemption from Institutional Review Board of St. Vincent’s Hospital (exemption number: VIRB-20220217-001).
Patient consent
The patient provided written informed consent for the publication and the use of his images.
Author contribution
Conceptualization: Jin Soo Lim. Data curation: Hyun Jung Ryoo. Writing - original draft: Hyun Jung Ryoo. Writing - review & editing: Hyun Jung Ryoo, Jin Soo Lim, Min Cheol Kim. Supervision: Min Cheol Kim. All authors read and approved the final manuscript.


