2. Jang YJ, Alfanta EM. Rhinoplasty in the Asian nose. Facial Plast Surg Clin North Am 2014;22:357-77.


3. Hwang NH, Han SK, Dhong ES. Adaptive planning and overcoming pitfalls in Asian rhinoplasty. Arch Aesthetic Plast Surg 2014;20:8-14.

4. Romo T 3rd, Pearson JM. Nasal implants. Facial Plast Surg Clin North Am 2008;16:123-32.


5. Parker Porter J. Grafts in rhinoplasty: alloplastic vs. autogenous. Arch Otolaryngol Head Neck Surg 2000;126:558-61.


8. Kim JH, Song JW, Park SW, Oh WS, Lee JH. 10th rib cartilage: another option of the costal cartilage graft for rhinoplasty. Arch Aesthetic Plast Surg 2015;21:47-53.

9. Rohrich RJ, Dayan E, Durand PD, Brito I, Gronet E. Warping characteristics of rib allograft cartilage. Plast Reconstr Surg 2020;146:37e-42e.


11. Kim IS. Augmentation rhinoplasty using silicone implants. Facial Plast Surg Clin North Am 2018;26:285-93.


12. Kangesu L, Goodacre TE, Stanley PR. Survival of irradiated glutaraldehyde preserved bovine cartilage in nasal reconstruction: a retrospective study. Br J Plast Surg 1991;44:483-5.


13. Lo S, Kenway B, Hosni A. Acellular porcine dermal collagen implant in saddle nose deformity. Plast Reconstr Surg 2011;128:82e-84e.


14. Das P, Singh YP, Joardar SN, Biswas BK, Bhattacharya R, Nandi SK, et al. Decellularized caprine conchal cartilage toward repair and regeneration of damaged cartilage. ACS Appl Bio Mater 2019;2:2037-49.


15. Velasquez N, Huang Z, Humphreys IM, Nayak JV. Inferior turbinate reconstruction using porcine small intestine submucosal xenograft demonstrates improved quality of life outcomes in patients with empty nose syndrome. Int Forum Allergy Rhinol 2015;5:1077-81.


16. Marianetti TM, Grussu F, Cervelli D, Gasparini G, Pelo S. Equine pericardium membrane to prevent dorsal irregularities in rhinoplasty. Ann Plast Surg 2014;73:128-30.


18. Jeong JY, Ha Y, Kim S, Yang HJ, Oh SH. Availability and safety of osteotomy in esthetic rhinoplasty of East Asian patients. Ann Plast Surg 2018;81:141-5.


19. Sena Esteves S, Goncalves Ferreira M, Carvalho Almeida J, Abrunhosa J, Almeida E, Sousa C. Evaluation of aesthetic and functional outcomes in rhinoplasty surgery: a prospective study. Braz J Otorhinolaryngol 2017;83:552-7.


20. DŌĆÖAgostino RB Sr, Massaro JM, Sullivan LM. Non-inferiority trials: design concepts and issues: the encounters of academic consultants in statistics. Stat Med 2003;22:169-86.


21. Ortiz-Monasterio F, Olmedo A, Oscoy LO. The use of cartilage grafts in primary aesthetic rhinoplasty. Plast Reconstr Surg 1981;67:597-605.


24. Gunter JP, Clark CP, Friedman RM. Internal stabilization of autogenous rib cartilage grafts in rhinoplasty: a barrier to cartilage warping. Plast Reconstr Surg 1997;100:161-9.


27. Ferrante B, Biessy R, Ducroz V, Gzaiel D, Leyder P. Correction of nose deviations using latero-septal grafts from irradiated bovine cartilage. Ann Chir Plast Esthet 1993;38:555-60.

28. Demir CI, Yasar EK, Davun K, Gok A, Uzun C, Alagoz MS. Creation of experimental human nose model with lyophilized and decellularized bovine cartilage xenograft. J Craniofac Surg 2018;29:1097-100.


30. von Bomhard A, Elsaesser A, Riepl R, Pippich K, Faust J, Schwarz S, et al. Cartilage regeneration using decellularized cartilage matrix: long-term comparison of subcutaneous and intranasal placement in a rabbit model. J Craniomaxillofac Surg 2019;47:682-94.


32. Benders KE, van Weeren PR, Badylak SF, Saris DB, Dhert WJ, Malda J. Extracellular matrix scaffolds for cartilage and bone regeneration. Trends Biotechnol 2013;31:169-76.


33. Stone KR, Walgenbach AW, Abrams JT, Nelson J, Gillett N, Galili U. Porcine and bovine cartilage transplants in cynomolgus monkey: I. A model for chronic xenograft rejection. Transplantation 1997;63:640-5.


34. Schwarz S, Koerber L, Elsaesser AF, Goldberg-Bockhorn E, Seitz AM, Durselen L, et al. Decellularized cartilage matrix as a novel biomatrix for cartilage tissue-engineering applications. Tissue Eng Part A 2012;18:2195-209.


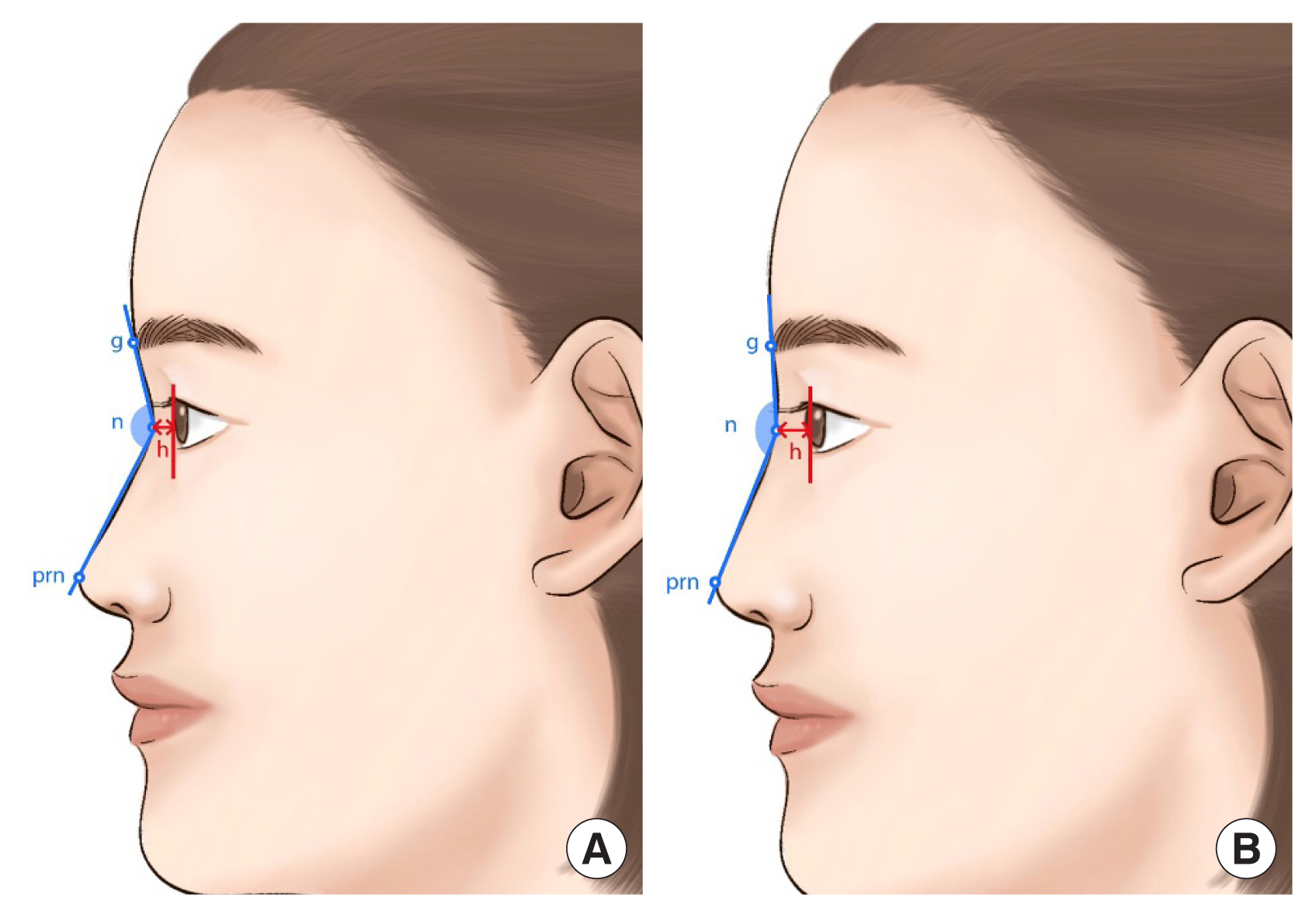
36. Wang JH, Jang YJ, Park SK, Lee BJ. Measurement of aesthetic proportions in the profile view of Koreans. Ann Plast Surg 2009;62:109-13.