|
 |
- Search
| Arch Craniofac Surg > Volume 24(2); 2023 > Article |
|
Abstract
Endotracheal intubation is often necessary in the course of animal experiments, especially in craniofacial surgery. However, endotracheal intubation can be a major burden in this context. The authors performed simple and cost-saving method using a 200 ╬╝L yellow micropipette tip, and the success of this method was demonstrated by X-ray and autopsy. We used a total of 30 rats. After the rats were fixed with a plaster, the trachea and vocal cords were visualized with the tongue pulled back. Under direct visualization of the vocal cords, a curving micropipette tip was advanced into the trachea. This method can be learned quickly and applied successfully by general experimenters. We successfully intubated all 30 rats without any complications. The success rate of micropipette tip intubation was 100%. This procedure was performed by one experimenter within 2 to 3 minutes after induction of anesthesia. We demonstrated its superiority by X-ray and autopsy. Herein, we describe endotracheal intubation of rats using micropipette tips. To the best of our knowledge, this method is novel and represents the simplest and most efficient means of intubation in rats, providing an alternative to conventional endotracheal intubation.
Rats are widely used in animal testing because they are easy to obtain and manage. Additionally, rats do not need large spaces or incur large costs. Most surgical experiments in rats require general anesthesia and some form of airway management, such as tracheostomy, endotracheal intubation or nasotracheal intubation. Nevertheless, given the anatomical complexity in the oral cavity of rats, it would be difficult to perform endotracheal intubation. Nasotracheal intubation is even more problematic. Unlike human airway management, rat airway management often requires tracheostomy due to the difficulty of endotracheal intubation. Therefore, an easy endotracheal intubation method is desperately required by many animal experiment laboratories in various academic areas. With a convenient method of intubation using a micropipette tip, rat experiments could be made easier for researchers in a range of academic areas; for example, the authors used this technique when conducting their study, ŌĆ£The healing of critical-sized bone defect of rat zygomatic arch with particulate bone graft and bone morphogenetic protein-2.ŌĆØ
Adult male SpragueŌĆōDawley rats aged 12 weeks and weighing 300 to 600 g each were housed in plastic cages in which standard chow and water were available. Animals were habituated to their environment for 3 to 6 days after arrival and were then fasted for 16 hours prior to procedures.
A total of 30 SpragueŌĆōDawley rats were studied for endotracheal intubation. Rats were anesthetized by intraperitoneal injection of a mixture of ketamine (50 mg/kg) and xylazine (4 mg/kg) [1], injected as a mixture of 0.1 cc/100 g of 2 cc ketamine (50 mg/mL) and 1.6 cc xylazine (4 mg/kg).
Before intubation, the rat was anesthetized with ether until the eyelid closure reflex was lost. After general anesthesia, the rats were laid down on a flat inclined board and suspended by a ribbon hooked around the upper incisors for better visualization and control. The tongue was fixed to one side [1,2]. One person intubated the animal while standing behind the inclined metal plate [1,2], and the other person stood to the side to help keep the ratŌĆÖs mouth open [1].
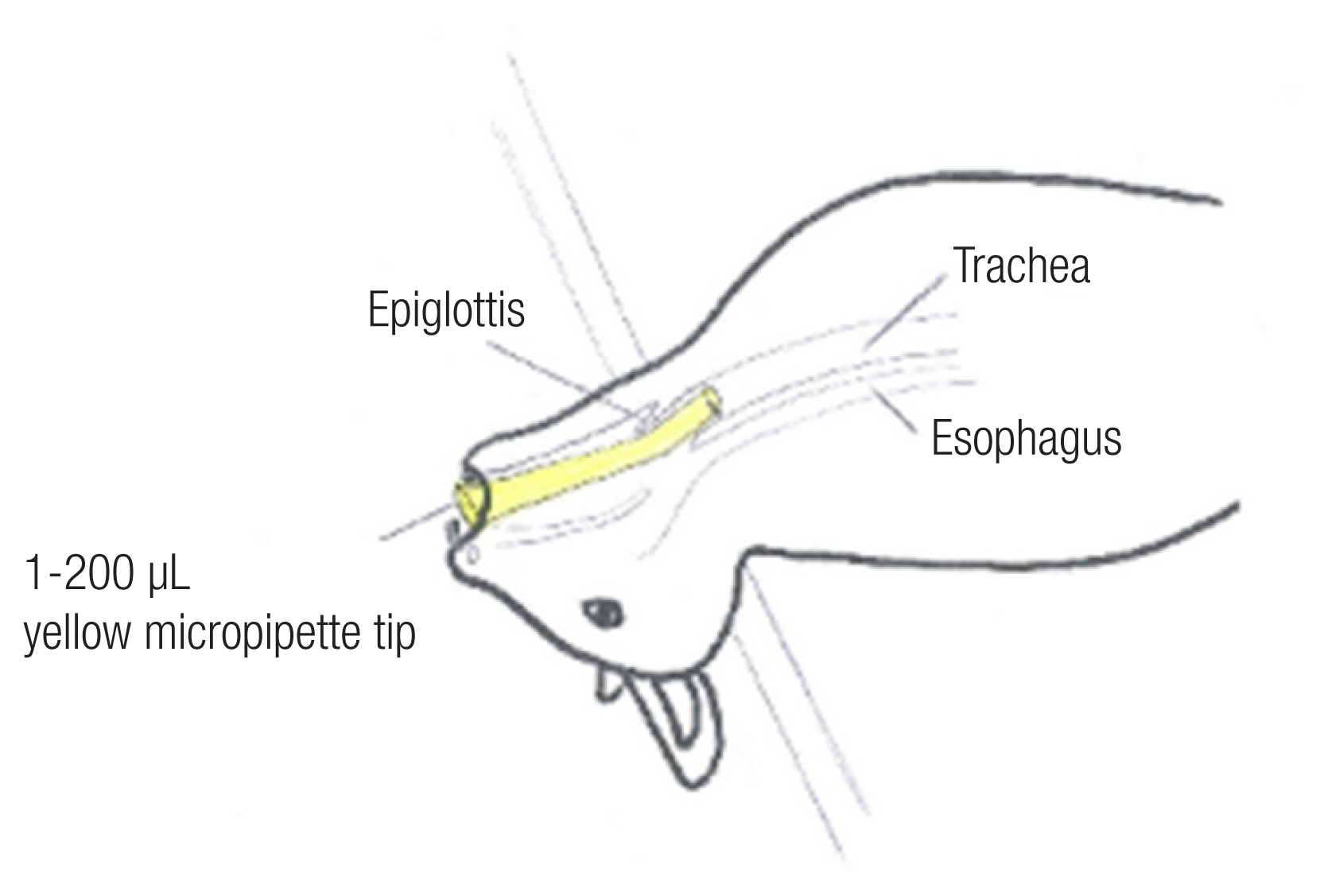
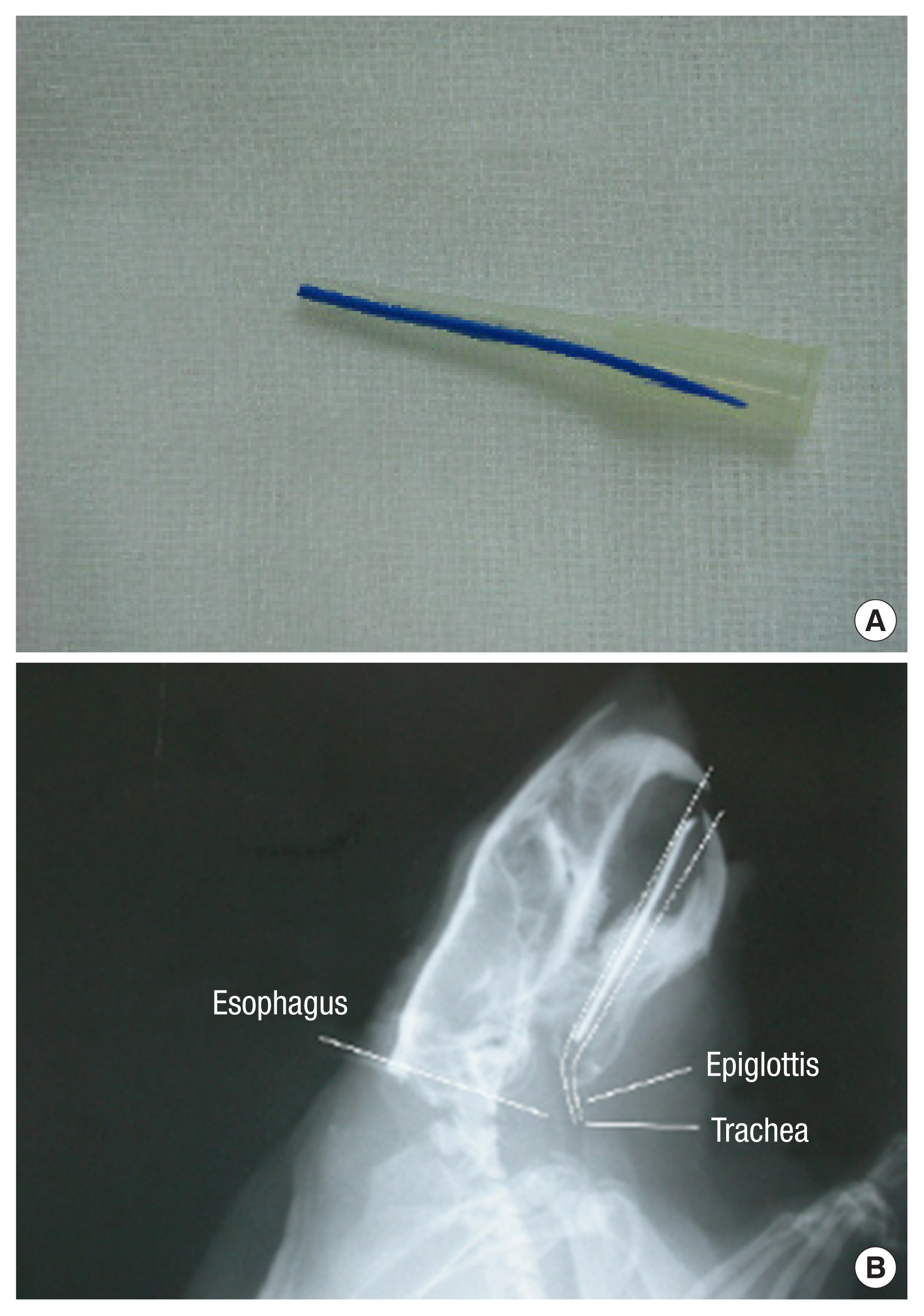
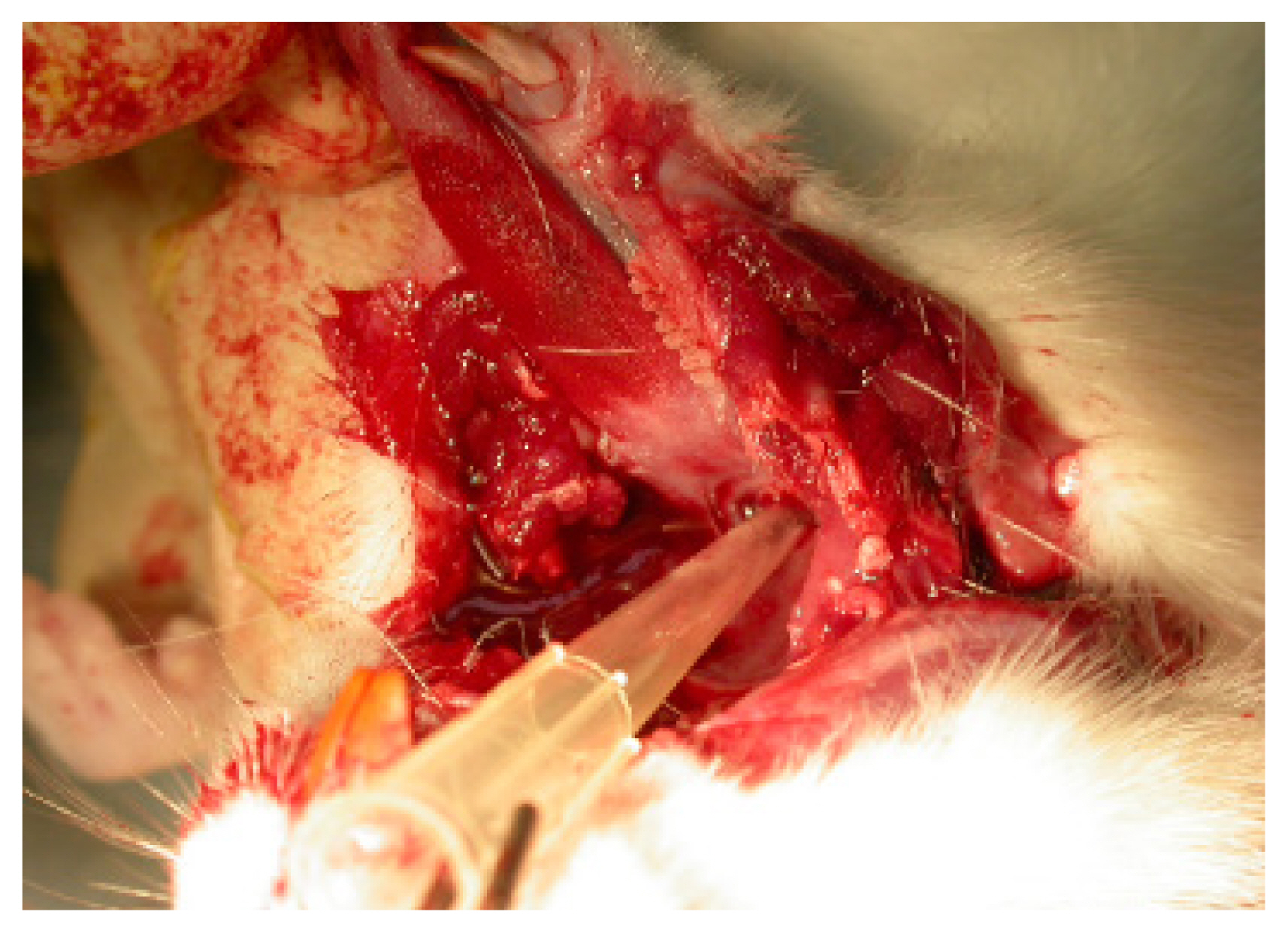
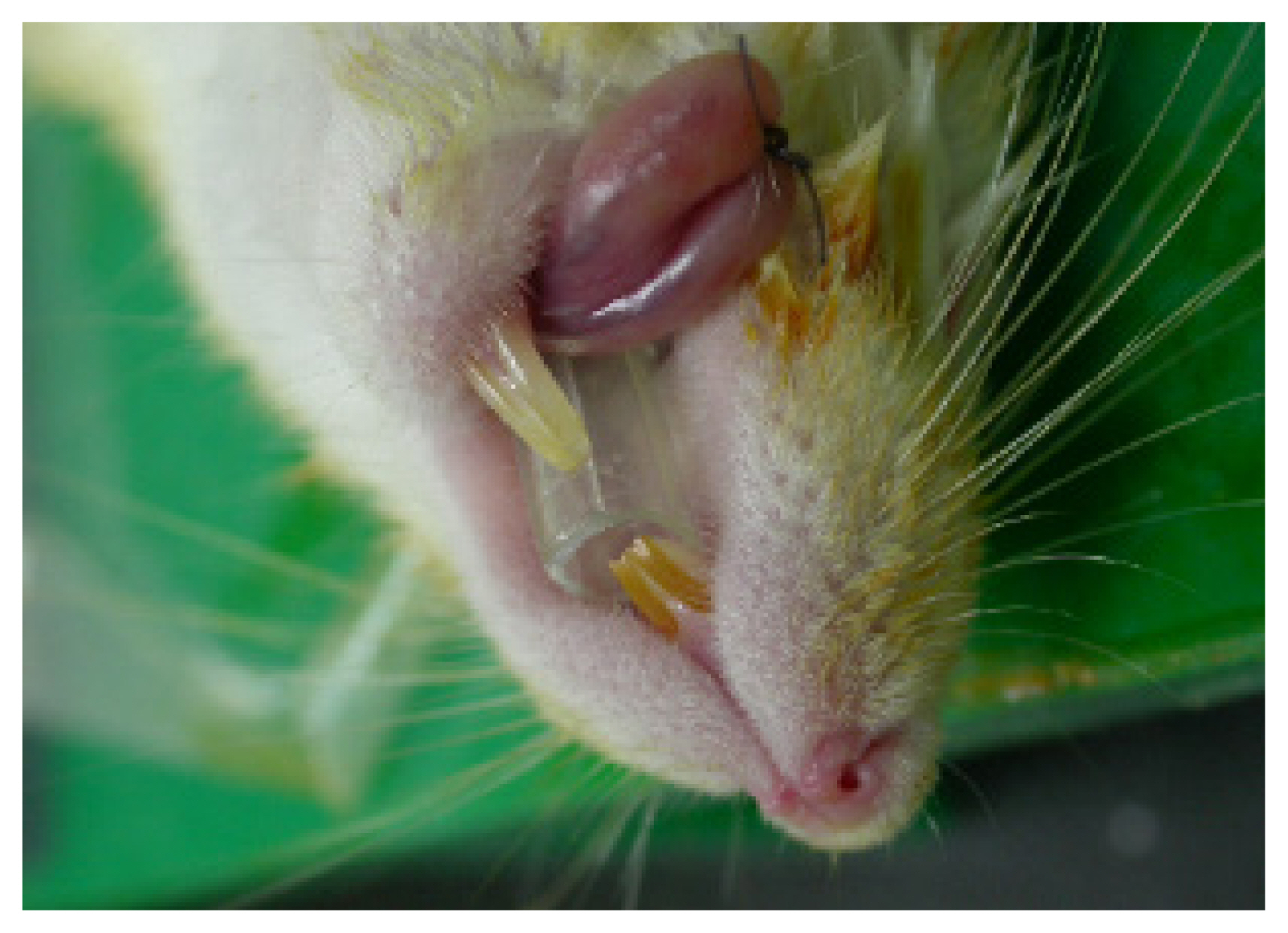
For intubation, we used a 200 ╬╝L yellow micropipette tip (AXYGEN, T-200-Y, length 5 cm with 7 mm caliber) (Fig. 1). The micropipette was cut to alter the diameter of the tip from 2 mm to 4 mm, which is the diameter of the ratŌĆÖs endotracheal tube, and intubation was performed in only a few minutes. The apex of the micropipette tip, approximately 0.6 cm in length, was bent 20┬░. The board was raised up to a 45┬░ angle, and light was shone on the mouth. Trachea and vocal cords were visualized with the tongue pulled back. Under direct vision of the vocal cords and the upper airways [1ŌĆō4]. The tip of the 200 ╬╝L yellow micropipette tip was removed for efficient airway passage, and a radiologic marker was attached to check the position for the experiment. For some cases, secretions were suctioned (Figs. 2, 3) [5,6]. Next, the airway was confirmed by X-rays (Fig. 4) and autopsy (Fig. 5) [7].
This technique can be performed within only 2 to 3 minutes, and anyone can successfully perform it after a very short learning period.
We intubated 30 rats using our method, and all 30 rats were successfully intubated without difficulty or mortality. One hundred percent of the animals were successfully intubated within 3 minutes. We confirmed this result by autopsy after euthanasia. No animals had laryngospasm or orotracheal trauma. Additionally, we confirmed successful intubation results with X-ray (Fig. 4) and autopsy (Fig. 5). Because micropipette tips are inexpensive and plentiful in the laboratory, this technique gives stability to rat experiments. The mean maintenance time of intubated status using a micropipette was 33.2 minutes (standardized deviation: 2.8). Additionally, this method prevents mortality in surgical experiments. After completion of the experiment, all rats were euthanized via anesthetic overdose.
In the current study, we have demonstrated micropipette intubation instead of a commercial endotracheal tube by a single operator. The reason to choose the micropipette instead of the intubation tube is that the micropipette is very soft and flexible. Additionally, the micropipette is one of the simple tools to obtain in a lab. When animals are deeply anesthetized or sedated, this method provides stable and reliable respiration to rats. Many other methods for rodent intubation have been described in the literature [3,7,8].
Vergari et al. [3] proposed in ŌĆ£Video-assisted orotracheal intubation in miceŌĆØ that straight fiber-optic arthroscopy is useful for endotracheal intubation. Clary et al. [7] also proposed in ŌĆ£Videoendoscopic endotracheal intubation of the ratŌĆØ that videoendoscopic endotracheal intubation is a simple and safe technique for endotracheal intubation in the rat that permits the introduction of a large-bore tube under direct visualization using equipment commonly found in the endo-surgical research setting. Ayoub et al. [9] proposed in ŌĆ£Transtracheal oxygenation*ŌĆØ that transtracheal oxygenation was as effective as conventional positive-pressure ventilation with 100% O2 for securing oxygenation, resuscitation, and short-term survival and more effective than O2 delivered through a mask. As described above, proper intubation is the most complicated and important thing in animal laboratories.
This technique is one of the simplest intubation methods for rats because video endoscopy, metal plates, 3-D printing, etc., are not needed. Micropipette tips are considered a cost-effective tool, especially in small labs that do not have an endoscope or rat endotracheal tube. If mouse is smaller than ours, 20 ╬╝L white tip (AXYGEN, T-300) instead of 200 ╬╝L yellow tip (AXYGEN T-200-Y) could be used. To the best of our knowledge, although other micropipette tips with various sizes have not been applied to other animal models, even the rats used in this study, we could not establish and standardize the modality of intubation in animal models. The maintenance time revealed in this study was relatively sufficient for our study; nevertheless, the maximum maintenance time for this modality of intubation was not confirmed in this study. Therefore, further studies are warranted to evaluate the possible maximum maintenance time of this procedure.
X-ray indicators are not recommended for all cases. Those are helpful for the novice and, also the experts especially to the maxillofacial surgeon on this method (Fig. 6). The authors expect that this micropipette intubation method will contribute to animal testing and further research and will be reported in conjunction with a review of the literature.
Notes
Ethical approval
This study was approved by the Dong-A Institutional University Animal Experimental Ethics Committee (DIACUC approval No. 22-39).
Author contributions
Conceptualization: Myung-Good Kim. Project administration: Myung-Good Kim, Dong Min Lee. Visualization: Jeong-Ho Ryu. Writing - original draft: Jeong-Ho Ryu, Tae-Seo Park, Ji-An Choi. Writing - review & editing: Jeong-Ho Ryu, Tae-Seo Park, Ji-An Choi. Investigation: Myung-Good Kim, Ji-An Choi. Resources: Myung-Good Kim, Dong Min Lee, Tae-Seo Park, Ji-An Choi. Supervision: Myung-Good Kim, Tae-Seo Park. Validation: Dong Min Lee, Ji-An Choi.
Fig.┬Ā1
(A) Unsuitability of a straight 200 ╬╝L yellow micropipette tip (AXYGEN, T-200-Y, length 5 cm with 7 mm caliber). (B) Preparation of the yellow tip by curving and sectioning to render it suitable for intubation.

Fig.┬Ā3
A 200 ╬╝L yellow micropipette tip was pushed carefully in the trachea, and the rat was confirmed to be breathing.
REFERENCES
1. Samsamshariat SA, Movahed MR. Using a 0.035-in. straight-tip wire and a small infant laryngoscope for safe and easy endotracheal intubations in rats for cardiovascular research. Cardiovasc Revasc Med 2005;6:160-2.


2. Kastl S, Kotschenreuther U, Hille B, Schmidt J, Gepp H, Hohenberger W. Simplification of rat intubation on inclined metal plate. Adv Physiol Educ 2004;28:29-32.


3. Vergari A, Polito A, Musumeci M, Palazzesi S, Marano G. Video-assisted orotracheal intubation in mice. Lab Anim 2003;37:204-6.



4. Cambron H, Latulippe JF, Nguyen T, Cartier R. Orotracheal intubation of rats by transillumination. Lab Anim Sci 1995;45:303-4.

5. Tran HS, Puc MM, Tran JL, Del Rossi AJ, Hewitt CW. A method of endoscopic endotracheal intubation in rabbits. Lab Anim 2001;35:249-52.



6. Corleta O, Habazettl H, Kreimeier U, Vollmar B. Modified retrograde orotracheal intubation technique for airway access in rabbits. Eur Surg Res 1992;24:129-32.


7. Clary EM, OŌĆÖHalloran EK, de la Fuente SG, Eubanks S. Videoendoscopic endotracheal intubation of the rat. Lab Anim 2004;38:158-61.