INTRODUCTION
Free tissue transfer has emerged as the preferred method for reconstructing head and neck defects, with a success rate of approximately 95% [
1,
2]. Despite the increasing success rate of these challenging and complex head and neck reconstructions procedures, the remaining cases of free tissue transfer failure still warrant attention because they cause substantial patient morbidity and diminish patientsŌĆÖ quality of life. Therefore, this single-institution study investigated the causes of flap failure, even though it occurs in a small percentage of cases.
The known risk factors for free flap failure can be classified as epidemiological factors, comorbidities, and other factors. The epidemiological risk factors include sex and age. In addition, peripheral vascular disease, coronary artery disease, diabetes, and hyperlipidemia have been reported as comorbidities that affect free flap failure [
3,
4]. Neo-adjuvant therapies such as irradiation and chemotherapy, anticoagulant use, and operative time are also risk factors for free flap failure in head and neck reconstruction [
5,
6].
In revision surgery performed for poor circulation to the flap, pedicle thrombosis was the most frequently reported cause of the circulation problem. Pedicle thrombosis can occur in both veins and arteries, but the incidence of venous thrombosis, which is mainly caused by kinking or twisting of the vein, is higher than that of arterial thrombosis. Arterial thrombosis has a lower incidence rate, but also a lower salvage rate. Arterial thrombosis causes flap failure via ischemia, and hematoma and bleeding have also been reported to lead to flap failure [
7-
10].
We performed a retrospective analysis of patients who underwent free tissue transfer during reconstruction for head and neck cancer between 2016 and 2021. The study sample included 60 cases of microvascular head and neck reconstruction performed by a single surgeon at a single institution. The aim of this study was to predict the surgical prognosis of free tissue transfers to head and neck defects, and the findings are expected to yield insights on how to improve the success rate by correcting modifiable risk factors.
RESULTS
During the study period, the overall number of patients was 58 and the total number of free flap procedures was 60; one patient underwent subsequent free flap surgery twice. The revision procedures were not performed from new donor tissue, and therefore were not counted as separate cases of surgery. Revision surgery (e.g., for re-anastomosis) was needed in 14 patients. Subsequent free flap surgery was performed in one patient, and three free flaps (5%) could not be salvaged. As shown in
Table 1, the flap outcomes showed a significant association with patientsŌĆÖ age. Patients with successful free flaps had a mean age of 64.41 years and those with successfully salvaged flap revisions were on mean of 58.10 years old, while cases of free flap failure had a mean age of 77.67 years. Four of 21 female patients needed revision surgery, which led to successful salvage in two cases and failure in two cases. The oral cavity was the defect location with the highest failure rate in the salvaged revision group, and the midface area had the highest failure rate in the flap failure group. Two of the 12 patients in the salvaged revision group and all three patients with failed flaps had hypertension. A coronary interventional procedure was performed in two of the three patients with failed flaps. Other medical comorbidities, such as diabetes and renal disease, did not show significant associations with flap outcomes.
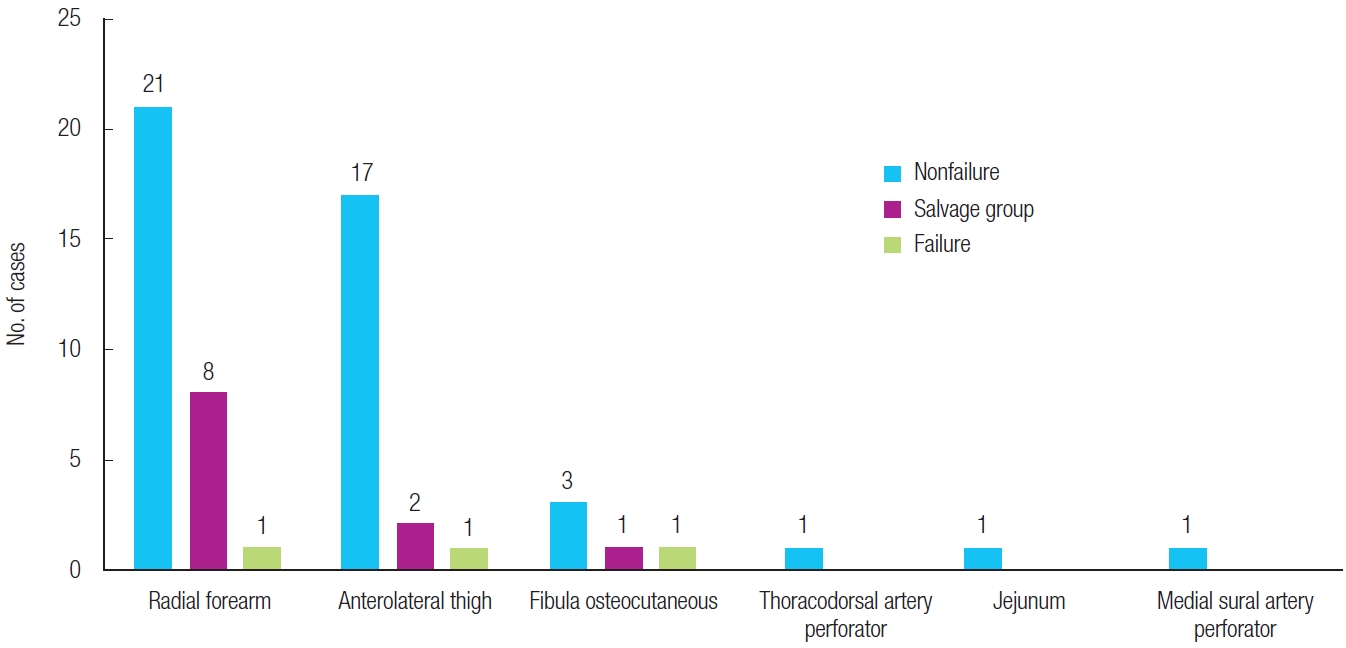
The type of flap performed and the success rate of each flap are presented in
Fig. 1. Radial forearm free flaps accounted for the largest proportion of cases, followed by anterolateral thigh (ALT) free flaps. One patient each in the flap failure group had a radial forearm free flap, ALT free flap, and fibula osteocutaneous free flap. Overall, ALT free flaps were most common (20 cases, 34%), followed by fibula osteocutaneous free flaps (4 cases, 7%).
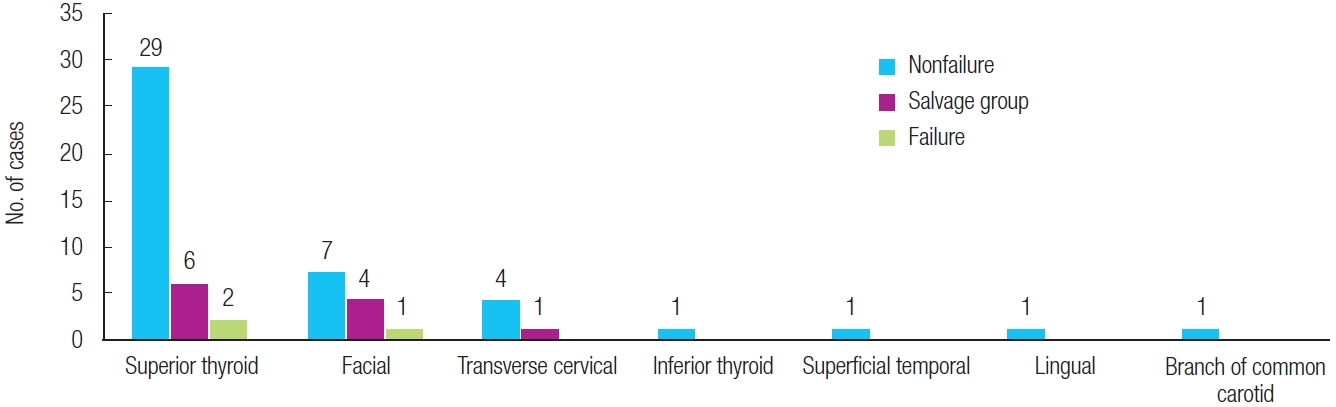
The superior thyroid artery was the most common recipient artery used for free tissue transfer, followed by the facial artery and the transverse cervical artery. In both the salvage group and the flap failure group, the superior thyroid artery was used most frequently (55% and 67%, respectively) (
Fig. 2).
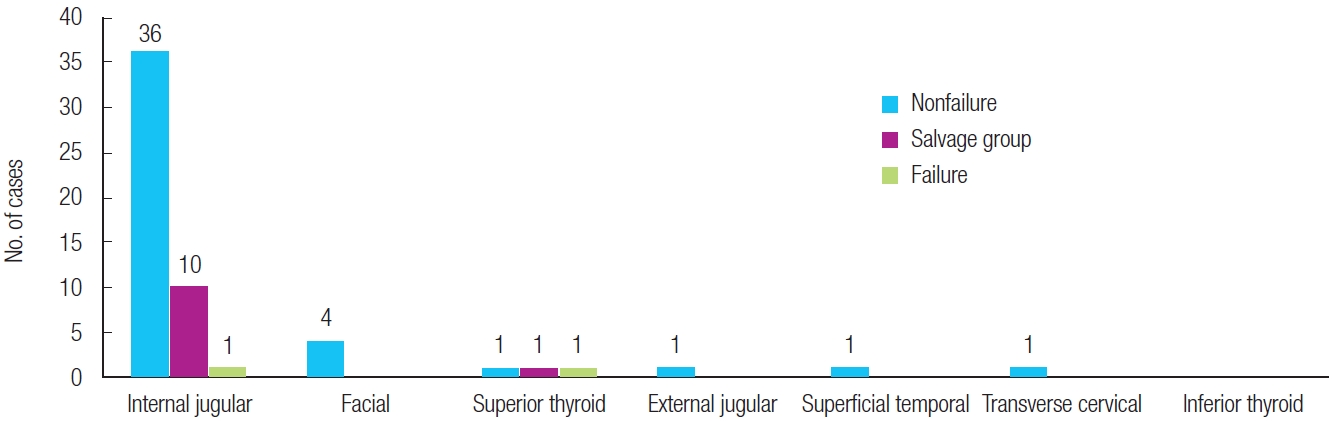
The internal jugular vein was the most common recipient vein in salvage cases, as it was used in 10 out of 11 cases in this group. In the flap failure group, the internal jugular vein, the superior thyroid vein, and the inferior thyroid vein accounted for one case each (
Fig. 3).
In one salvage case, re-anastomosis was performed by changing the recipient artery to another artery on the same side. In another case, re-anastomosis was performed by changing from end-to-end to end-to-side anastomosis in the recipient vein (internal jugular vein). Systemic heparinization was conducted to prevent thrombosis.
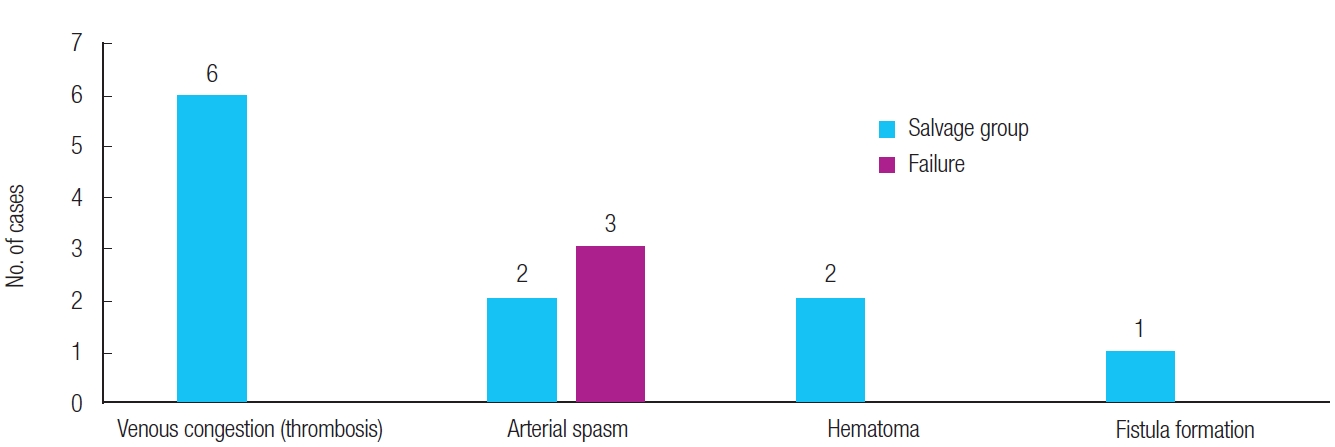
In total, 14 cases required revision surgery. Venous congestion/thrombosis (6 cases, 55%), arterial spasm (2 cases), and hematoma around the skin closure site rather than in the vessel lumen (2 cases) were the most common reasons in the salvage group. Revision was performed for arterial spasm in three cases in the flap failure group, but the flap was lost (
Fig. 4).
DISCUSSION
Several factors influence the failure of free flaps used in head and neck reconstruction procedures. We investigated possible risk factors, including age, sex, defect location, and comorbidities (diabetes, cardiovascular disease, hypertension, renal disease, smoking, alcohol use, and history of radiotherapy and chemotherapy). Several other studies have identified additional potential risk factors, such as peripheral vascular disease, hyperlipidemia, American Society of Anesthesiologists classification, operative time, and anticoagulant medications such as aspirin, clopidogrel, and low-molecular-weight heparin [
5,
6,
11-
14]. The present study analyzed 58 patients who underwent head and neck reconstruction with free flaps. Age was a significant factor affecting the success or failure of free flaps in our patient group. Population aging has been accompanied by an increasing number of elderly patients requiring head and neck reconstruction. The development of microsurgery has also allowed more elderly patients to become candidates for free flap surgery. In our patient sample, the mean age of patients with successfully salvaged free flaps was 63.15 years, while it was 77.67 years in those with failed free flaps. The mean age difference between the two groups was 14.52 years. Medical complications were not considered in this study. However, Jubbal et al. [
15] reported that surgical complications in microvascular free flap surgery in elderly patients occurred with equal frequency across all groups, but the medical complication rate increased significantly with age. The results for sex were not statistically significant; two out of the three free flap failures were in female patients and one was in a male patient.
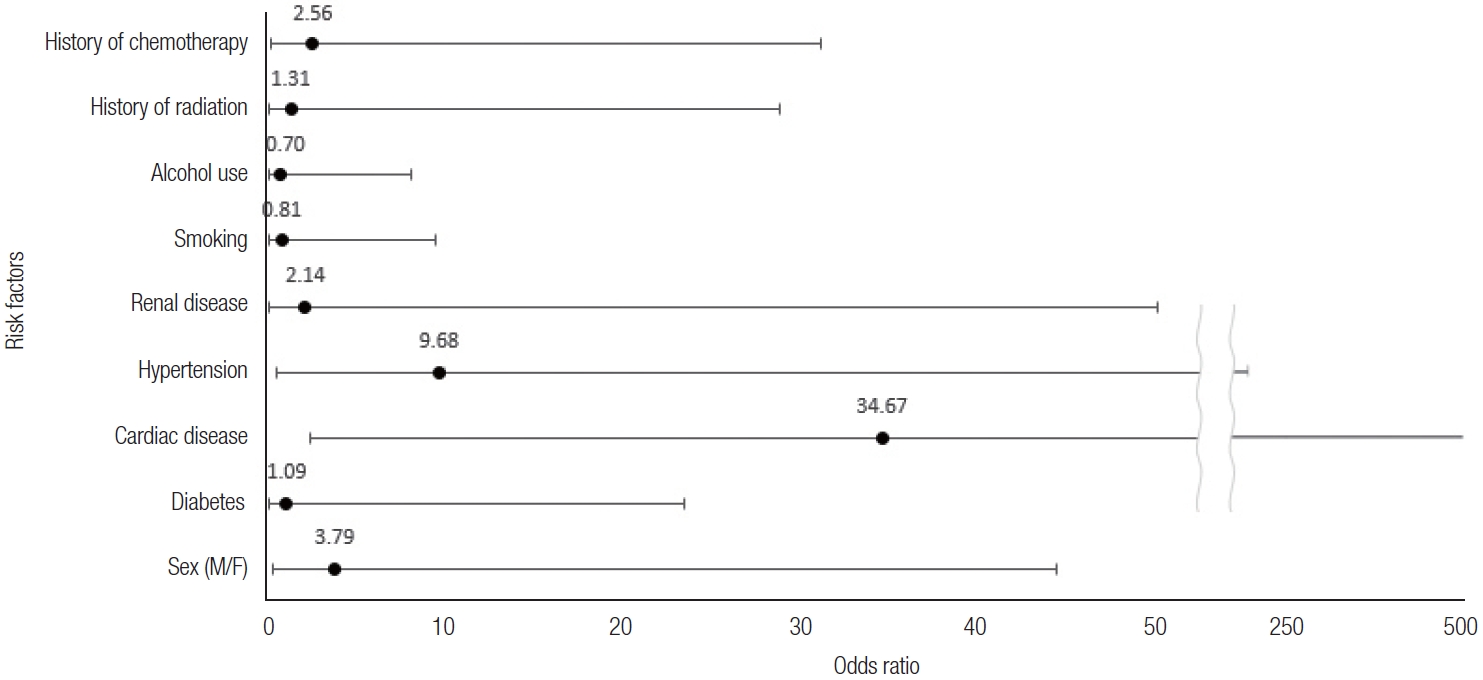
Cardiovascular disease was a significant factor affecting flap failure in our patient group (
Table 2,
Fig. 5). Cardiovascular disease is a broad category encompassing various conditions that affect the heart and blood vessels, and in this study, we considered acute myocardial infarction (AMI), angina, myocarditis, arrhythmia, atherosclerosis, peripheral arterial occlusive disease, aneurysm, hypertension, and vasculitis. The conditions present in our patients were hypertension, arrhythmia, AMI, and peripheral arterial occlusive disease. Although not directly addressing the effect on surgical flaps, a study by Simpson et al. [
16] reported that a history of AMI was significantly associated with mortality and stroke. According to that study, increased metabolic demands on the heart due to fluctuations in blood pressure and the secretion of stress hormones, which increase the cardiac workload, can cause AMI. Among our patients with a history of cardiovascular disease, these effects can cause further changes in an already unstable cardiovascular system and hemodynamic status and lead to failure in a microvascular free flap surgery. In a study by Jan et al. [
17], the blood viscosity of patients with AMI was measured and, although appropriate interventional and medical treatments were performed, the blood viscosity remained high in the AMI patient group. A study by Lenz et al. [
18] confirmed that blood viscosity can affect tissue perfusion. Thus, elevated blood viscosity in patients with AMI is likely to degrade tissue perfusion and contribute to free flap failure.
In patients with pre-existing cardiovascular disease who experienced flap failure, it was sometimes challenging to maintain stable blood pressure during and after surgery. Arterial spasm could have occurred, considering the possibility of thrombus formation in the blood vessels and the consequences of unstable blood pressure. The intraoperative use of norepinephrine to maintain appropriate blood pressure could potentially be associated with arterial spasm, but norepinephrine was not used in the three cases of flap failure. Nonetheless, our findings should be interpreted with caution due to the limited number of cases of flap failure, which may restrict the generalizability and statistical significance of our findings.
There were six flap types in our study when classified by donor site, and the type of flap was not associated with flap failure. However, a higher rate of flap revision was observed in cases of radial forearm free flaps, although this finding was not statistically significant. An explanation for this may be that, in the early stages when the author performed free flaps, the author predominantly used the radial forearm as the donor site, and there may have been issues with microsurgical techniques. The microsurgical techniques of an inexperienced operator can include performing anastomosis under tension on blood vessels or encountering vascular kinking due to subtle positional changes in the internal jugular vein after end-to-side anastomosis during surgery. In addition, there was no significant difference in the probability of flap failure according to the recipient arteries or recipient veins used. This was also confirmed by the study by Bui et al. [
10], which analyzed the revision of free flaps due to vascular thrombosis in 1,193 free flap cases. According to that study, the most important factor affecting the prognosis of a free flap was avoiding anastomosis in areas with a history of previous trauma, radiation, or extant disease, not the specific artery or vein used.
In our study, venous congestion was the most common reason for revision surgery, followed by arterial spasm, hematoma, and fistula. According to a study by Boissiere et al. [
19] on flap venous congestion and salvage techniques, venous congestion can be broadly divided into extrinsic (mechanical) and intrinsic (microscopic) causes. Extrinsic causes include compression caused by kinking and twisting of the vessel, compressive hematoma due to skin closure with tension, and patient positioning problems that affect the pedicle of the flap. Venous congestion caused by kinking and twisting of the vessel can be prevented during surgery by marking the neutral site and shortening the redundant segment during anastomosis. In addition, the use of anchoring sutures to create a smooth curve can help prevent kinking of the pedicle [
19]. When performing skin closures, loose sutures are recommended so that pressure is not applied to the pedicle area. If loose suturing is impossible due to a lack of remaining skin, a rotation flap can be considered. Even after surgery, the patientŌĆÖs position, especially neck motion, should be carefully monitored so as not to compress the area. In most cases, emergency reoperation is required when congestion of the flap is suspected. For a thrombus in the pedicle, reanastomosis is recommended after performing a thrombectomy using a Fogarty catheter. If blood flow does not return, a nearby recipient vein must be located to perform a new anastomosis [
20-
22]. Venous congestion was the cause of most flap failures in our patients. However, the success rate of revisions for this cause was high. According to Fang et al. [
23], when an orocutaneous fistula (OCF) occurs, it can delay the initiation of adjuvant therapy and increase costs for the patient [
24]. Permanently impaired swallowing can ensue in rare cases. An OCF can usually be resolved with simple wound care, and in our patient with OCF, the affected area was closed after revision surgery with a non-microvascular procedure.
Arterial spasm occurred in five of our patients. Two out of 11 patients (18%) in the salvage group required flap revision due to arterial spasm. In addition, flap revision was required for all three cases in the free flap failure group with arterial spasm. Arterial spasm was the complication with the most negative impact on flap viability in our patient group. All three cases of flap failure were in patients aged 75 years or older. Due to the prolonged operative time (over 12 hours) and unstable blood pressure, postoperative intensive care unit management was implemented. In one case involving hypopharynx reconstruction, monitoring was not conducted. In the other two cases, arterial spasm was recognized at 4 hours and 3 days after surgery, respectively, and immediate revision surgery was deemed necessary. However, these patientsŌĆÖ general condition was not favorable, preventing us from proceeding with revision surgery. According to Rosette et al. [
25], who investigated the occurrence of arterial spasm during free flap surgery, the main reason why vasospasm occurs at the anastomosis site during microvascular surgery is the small diameter of the recipient artery. In addition, excessive manipulation of the vessel can traumatize it and induce vasospasm. Therefore, vasospasm can be prevented by (1) changing from end-to-end anastomosis (performed in 89% of procedures) to end-to-side anastomosis on a large vessel; (2) minimizing trauma to the recipient and flap arteries by performing atraumatic dissection and smooth manipulation of the vessels during anastomosis; and (3) avoiding anastomosis using a pathological artery that has been damaged by previous trauma or disease. In addition, men > 50 years of age and active smokers were more likely to develop arterial spasm. According to Hagau and LongroisŌĆÖs study of anesthesia in free flap patients [
26], smoking cessation 4 to 6 weeks prior to surgery can minimize the risk of vasoconstriction and flap compromise. It is also known from the work of Burkhard et al. [
27] that maintaining hemodynamic status is important in preventing arterial spasm and maintaining flap perfusion. Thus, maintaining proper blood pressure during free flap surgery, anesthesia, and after surgery is important for preventing arterial spasms.
We analyzed the failure of free flaps in patients who underwent head and neck reconstruction. Most previously published studies have focused on flap congestion caused by vein thrombosis, which is the most common vascular complication, but our cases presented a different perspective. In our cases, congestion was promptly detected during flap monitoring and could be easily resolved. In contrast, identifying arterial spasm is more challenging, and the success rate of revision surgery is lower [
28,
29]. Considering the small number of cases, it was difficult to generalize the cause of free flap failure, but statistically, the factors related to the failure of free flap surgery were relatively clear. Close postoperative monitoring for medical complications in elderly adult patients is important when performing free flap surgery, since cardiovascular disease was shown to be a statistically significant risk factor for flap failure. Attentive perioperative care is also required to prevent postoperative flap complications. All patients with free flap failure had arterial spasm, and careful attention should be paid to surgical technique and blood pressure management in order to prevent arterial spasm.