|
 |
- Search
| Arch Craniofac Surg > Volume 24(4); 2023 > Article |
|
Abstract
Rhabdomyosarcoma is the most common soft tissue sarcoma in children, accounting for 4.5% of all cases of cancer in childhood. Although the head and neck are the most common sites of rhabdomyosarcoma, oral lesions are relatively rare and account for only 10% to 12% of head and neck rhabdomyosarcoma cases. This is a case report of a girl aged 2 years and 1 month who initially presented with an upper lip mass that invaded the oral mucosa, oral skin, and nostril skin, causing narrowing of the airway. Through our case, we show that rapidly growing small round cell malignancies, especially rhabdomyosarcoma, can be effectively diagnosed and treated at the same time using primary resection with intraoperative frozen section biopsy and that the time spent waiting for the results of preoperative biopsy can be saved in this way, particularly when the patientŌĆÖs symptoms are intensifying rapidly and require immediate operation.
Rhabdomyosarcoma, the most common soft tissue sarcoma in children, accounts for a plurality of childhood cancer at up to 4.5% of all cases, and approximately 350 cases of rhabdomyosarcoma are diagnosed annually in the United States [1,2]. Rhabdomyosarcoma is an aggressive skeletal muscle malignancy of mesenchymal origin and is classified as a small round cell tumor [3]. Although the head and neck are the most common sites of rhabdomyosarcoma (35%), oral lesions are relatively uncommon and account for only 10% to 12% of head and neck rhabdomyosarcoma cases [4]. While many factors, including patient age and the location, size and resectability of the primary tumor, must be considered in order to select the proper treatment option for rhabdomyosarcoma, primary resection or at least open incisional biopsy is one of the key aspects of managing rhabdomyosarcoma [1]. Here, we present a case study illustrating the usefulness of simultaneous resection and diagnosis with intraoperative frozen section biopsy of rapidly growing orofacial small round cell tumors in children.
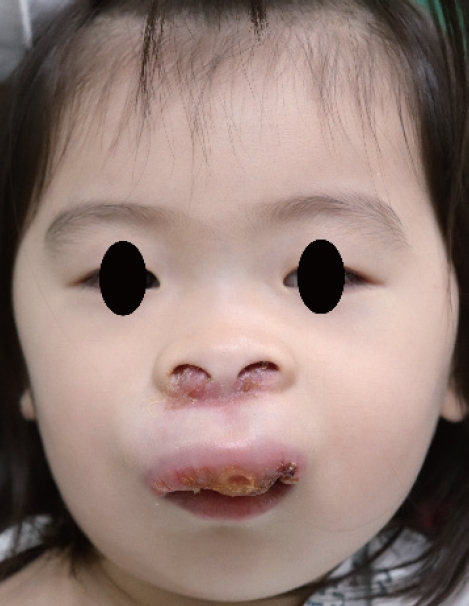
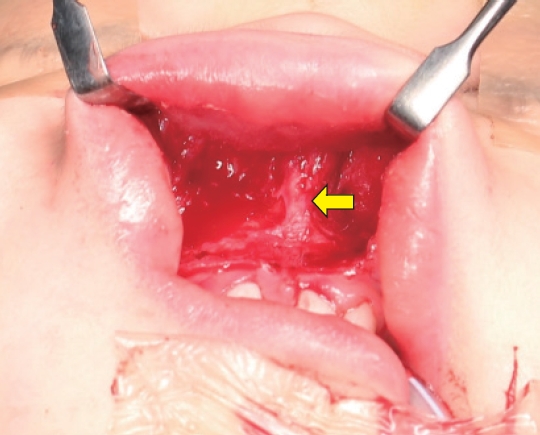
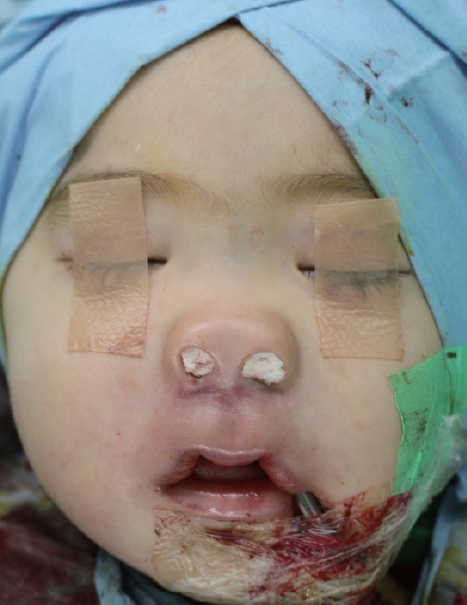
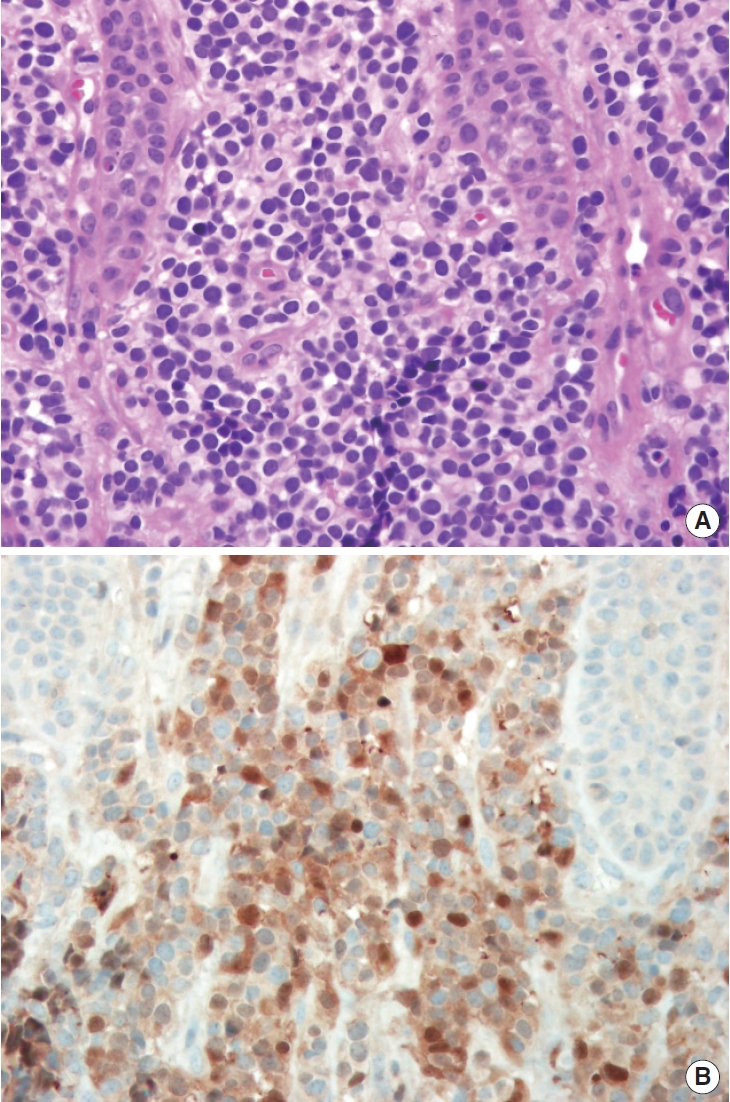
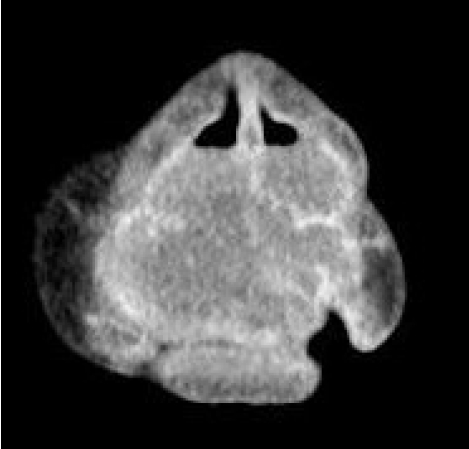
The patient was a girl aged 2 years and 11 months who had no medical history. She initially presented to our department with a mass of the upper lip, which had first been recognized by her parents 3 months ago. The lesion had been limited to the upper lip at first observation, according to her parents, but it had invaded the oral mucosa as well as the oral skin and nostril base, narrowing the airway, by the time she presented to our department (Fig. 1). We had strong suspicion about malignancy because of the pace of growth, and a computed tomography scan was performed immediately. Emergency reading of the computed tomography scan was conducted by a radiologist, and the results showed a poorly defined, heterogeneous and space-occupying tumor. The mass was relatively isodense with the adjoining muscle and partially occluded the airway (Fig. 2). As the computed tomography scan result implied a high likelihood of malignancy, we immediately decided to perform tumor resection with intraoperative frozen section biopsy in 1 week. The patient was hospitalized the day before the operation. The tumor had grown rapidly for a week since her last visit, and the patient seemed to have greater difficulty breathing than a week ago owing to the progressive narrowing of the airway (Fig. 3). Under general anesthesia, intraoperative frozen section was obtained. The result was small round cell malignancy. The pathologist advised us to excise as much of the tumor as possible, and we resected nearly the entire small round cell malignancy (Fig. 4). To secure the safety margin, it was confirmed that there was no invasion of tumor in the resection margin through four times of consecutive frozen section procedures during the operation. After resection of tumor, bilateral facial artery musculomucosal flap was performed intraorally to reconstruct the damaged nasal floor, oral mucosa and orbicularis oris muscle, and the inner lip was reconstructed with a primary repair (Fig. 5). As the precise histological examination of the obtained specimen showed small round cells that were strongly positive for the muscle-specific proteins actin, desmin and myoglobin, a final diagnosis of rhabdomyosarcoma was made 4 weeks after the operation (Fig. 6). The rhabdomyosarcoma corresponds to stage 3 on the TNM system. The patient is currently undergoing 8 weeks of chemotherapy under the guidance of a pediatric oncologist with a regimen of vincristine, dactinomycin and cyclophosphamide. She is also slated to receive adjuvant radiation therapy. The problem of respiratory function was completely solved and there was a significant improvement in appearance. During the 3-month follow-up period, there was no evidence of recurrence of rhabdomyosarcoma so far and regular outpatient follow-up is planned to surveil the relapse.
Anatomically, rhabdomyosarcoma of the head and neck can be categorized into two groups: parameningeal rhabdomyosarcoma including the nose, nasopharynx, paranasal sinus and middle ear and non-parameningeal rhabdomyosarcoma including the scalp, orbit, oral cavity and oropharynx [4]. Additionally, there are two major histologic subtypes of rhabdomyosarcoma: embryonal rhabdomyosarcoma, which typically arises in head and neck or genitourinary sites, and alveolar rhabdomyosarcoma, which occurs in the trunk and extremities [5]. In addition to histologic analysis, there are two staging classifications to stratify patients with rhabdomyosarcoma into risk-based treatment groups: the TNM system and the clinical group. Especially, the TNM staging assigns one of four stages based on the size and site of primary lesion, nodal involvement and presence of metastatic disease. The tumors that start in favorable sites including the eyelid and orbit, non-parameningeal head and neck sites and nonbladder, non-prostate genitourinary site with any size without distant metastatic lesions are classified into stage 1. Tumors less than 5 cm, which occur in unfavorable sites including the bladder, prostate, extremities, parameningeal sites and trunk without metastasis are classified into stage 2. Tumors more than 5 cm in size that had occurred in unfavorable sites are in stage 3. Also, tumors not more than 5 cm in size which had spread to nearby lymph nodes are in stage 3. Tumors with distant metastasis are assigned into stage 4 [6].
Once rhabdomyosarcoma is highly suspected, although biopsy for confirmation of histology is required when complete surgical excision is not available, primary surgical resection with reconstruction should be executed if the site and size of the tumor permit removal of the entire gross tumor without anatomical or functional defects [7-9]. Additionally, all rhabdomyosarcoma patients need chemotherapy with a backbone of vincristine, dactinomycin and cyclophosphamide, and radiotherapy is applied in almost all patients to enhance the outcome [1]. In some cases, it has been reported that rhabdomyosarcoma is associated with the malignant transformation of neurofibromatosis type I [10]. While the development of treatment over the last several decades has considerably improved the long-term survival rate, an estimated 25% to 30% of patients with localized disease still suffer relapses. In patients with distant metastasis, the recurrence rate approaches 70% [11].
In our case, the patient showed rapid growth of the orofacial mass with invasion of the skin at the bases of the nostrils, which caused narrowing of the airway that necessitated surgical excision, and the results of the computed tomography scan suggested a high probability of soft tissue malignancy. Therefore, we immediately decided not to carry out a separate biopsy procedure because it would take too long to obtain the results. Instead, we performed immediate surgical excision with an intraoperative frozen section procedure. It took only 20 minutes to obtain the diagnostic results, although more precise pathological examination with immunohistochemical staining was still required postoperatively.
Our case report illustrates that small round cell tumors showing rapid growth in size, especially rhabdomyosarcoma, can be effectively diagnosed and managed at the same time with primary resection in combination with intraoperative frozen section biopsy, and the time spent waiting for results from a preoperative biopsy can be saved in this way, especially when the patient has rapidly deteriorating symptoms that necessitate surgery, such as airway obstruction or proptosis.
Notes
Ethical approval
The study was approved for exemption by the Institutional Review Board of Inje University Ilsan Paik Hospital (IRB exemption No. ISPAIK 2021-05-016).
Patient consent
The patientŌĆÖs guardian provided written informed consent for the publication and the use of patient images.
Author contributions
Conceptualization: Youngwoong Choi, Soo Hyang Lee. Data curation: Ki Pyo Sung. Project administration: Soo Hyang Lee. Writing - original draft: Youngwoong Choi, Ki Pyo Sung. Writing - review & editing: Soo Hyang Lee, Ki Pyo Sung. Investigation: Youngwoong Choi, Soo Hyang Lee, Ki Pyo Sung. Supervision: Soo Hyang Lee.
Fig.┬Ā1.
A 2-year-old girl with a mass in the upper lip invading the oral mucosa, oral skin and the bases of nostrils.
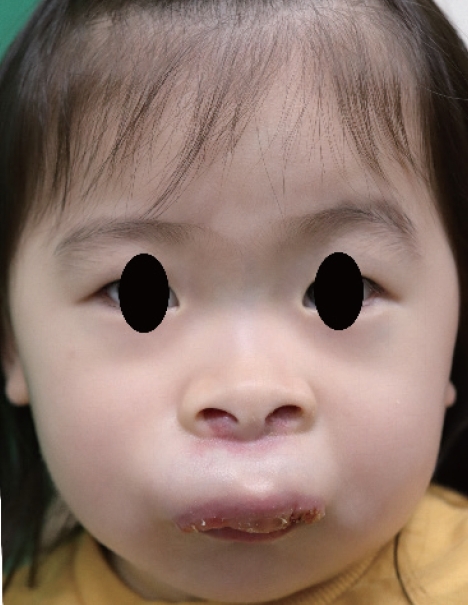
Fig.┬Ā2.
Preoperative computed tomography showing a poorly defined, heterogeneous orofacial tumor occupying the nasal cavity.
REFERENCES
2. Borinstein SC, Steppan D, Hayashi M, Loeb DM, Isakoff MS, Binitie O, et al. Consensus and controversies regarding the treatment of rhabdomyosarcoma. Pediatr Blood Cancer 2018;65:e26809.


4. Otmani N, Khattab M. Advanced orofacial rhabdomyosarcoma: a retrospective study of 31 cases. Int Arch Otorhinolaryngol 2016;20:207-11.



5. Rhee DS, Rodeberg DA, Baertschiger RM, Aldrink JH, Lautz TB, Grant C, et al. Update on pediatric rhabdomyosarcoma: a report from the APSA Cancer Committee. J Pediatr Surg 2020;55:1987-95.


6. Crist WM, Anderson JR, Meza JL, Fryer C, Raney RB, Ruymann FB, et al. Intergroup rhabdomyosarcoma study-IV: results for patients with nonmetastatic disease. J Clin Oncol 2001;19:3091-102.


7. Kim HS, Chung CH, Chang YJ. Free-flap reconstruction in recurrent head and neck cancer: a retrospective review of 124 cases. Arch Craniofac Surg 2020;21:27-34.




8. Koh IS, Sun H. A practical approach to nasal reconstruction in Asian patients. Arch Craniofac Surg 2021;22:268-75.




9. Lee Y, Lee D, Yeo H, Park H, Park H. Extraordinarily aggressive cutaneous sarcomatoid squamous cell carcinoma of the face: a case report. Arch Craniofac Surg 2022;23:77-82.




- TOOLS
-
METRICS

-
- 2 Crossref
- Scopus
- 1,211 View
- 37 Download
- Related articles in ACFS